Main group halides
From 1st and 2nd year lectures you discovered that the BH3 molecule is electron deficient and prefers to form dimers B2H6 which are unusal because of the bridging H-atoms. You also found out that a similar case exists for other group 13 elements. (As well as group II elements, for example Grinard reagents) In this project you will investigate the conformers and MOs of Al2Cl4Br2
- determine the five possible isomers and identify the symmetry of each isomer of Al2Cl4Br2
- compute the energy of the isomers with (a) 2 bridging Br ions and (b) the isomer with trans terminal Br and bridging Cl ions
- determine the relative energy of these isomers in kJ/mol
- discuss the relative stability of these conformers with respect to the bridging ions
- determine the dissociation energy for the lowest energy conformer into 2AlCl2Br
- is the product more or less stable than the isolated monomers?
- carry out a MO calcualtion on the lowest energy isomer (only!)
- visualise all the occupied valence MOs and the lowest 5 unoccupied MOs, you do not need to reproduce them all. If you don't know what the valence MOs are ask!
- in your wiki present 3 MOs ranging from highly bonding to highly antibonding (from any of the MOs you have visualised). Pick only occupied MOs. Draw a LCAO MO diagram of these orbitals (in chemdraw). Describe and annotate the interactions occurring in the MOs, identify the overal MO character. Refer to your MO course notes for the format of this description.
- here is the kind of thing you should produce, this is an example from an old exam
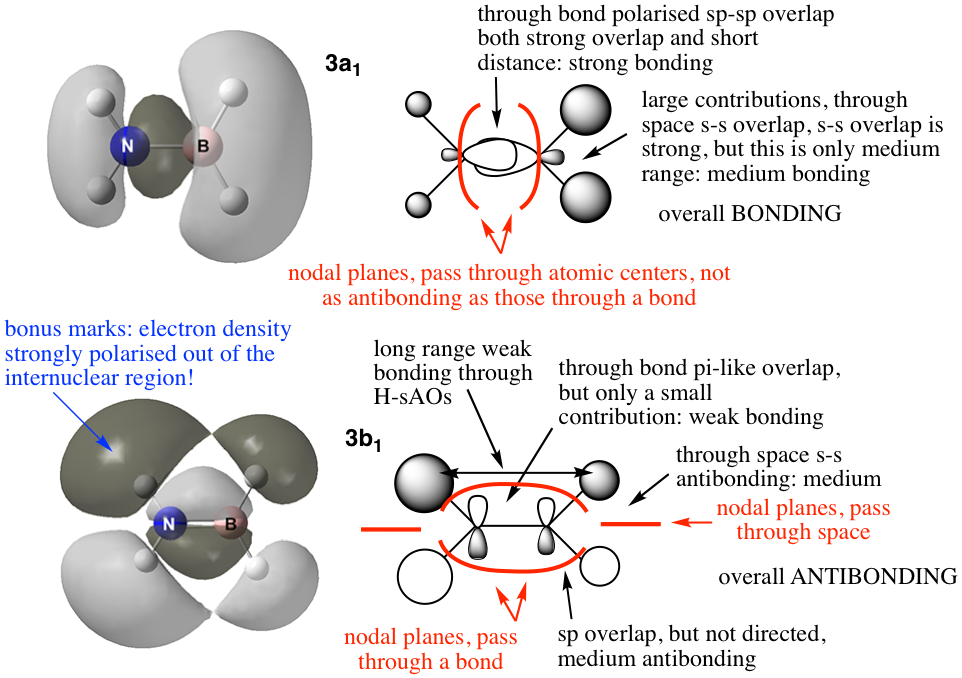
- I am looking for comments and annotations:
- indicating directly bonded vs through space interactions
- the distance dependence ie weak vs medium vs strong interactions
- discussion of polarized or directed interactions
- mention of s,p or d relative intensity of overlap
- the nodal character, on atoms is less antibonding than between atoms
- AND a clean tidy diagram that is well annotated
- optional Comment on or analyse or describe some aspect of this system which you found interesting.
- I am looking for comments and annotations:
