Aromaticity
Aromaticity is much more than the simplistic description you have so far experienced!
- Take a look at this article on double aromaticity, involving main group elements like Se "Double aromaticity arising from σ- and π-rings", Nature communications, 2018
- Or this article, using MOs and PES to establish aromaticity in B8- and B9- clusters, Angew. Chem. Int. Ed., 2003, article
- What about finding the smallest aromatic ring system: "The littlest aromate", C&EN, 2016.
- Scientists are people too and they argue about definitions, aromaticity is a divisive topic! "Aromaticity for all", C&EN, 2015.
You are all very familar with benzene as an aromatic compound. But how does this relate to the MO picture of molcular bonding? What happens when we exchange some of the "C-H" units by isoelectronic fragments containing main group elements. In this project you will investigate the MOs of benzene and its iso-electronic analogue borazine.
- optimise, and carry out a frequency analysis to confirm you have a minima of benzene and borazine.
- compute the MOs and carry out an NBO charge analysis (be sure you have NBO charges, not Mulliken!) for both structures. When you selected NBO (under "frequency analysis") you turned on the NBO charge analysis. Under "Results" select charge analysis and then under Type choose NBO. Play around with the options. You can then select colour and/or show numbers. Make sure you use the same colour range for both molecules when you save an image
- Compare the charge distribution for benzene and borazine, placing images side by side is not sufficient, list and discuss the charges. The key words here are "compare" and "discuss" just presenting the tables is not sufficient, you must interpret your results.
- visualise all the occupied valence MOs and the lowest 5 unoccupied MOs of both compounds, you do not need to reproduce them all. If you don't know what the valence MOs are ask!
- Note: almost all textbooks include only the pi orbitals when they discuss benzene, but as you can see there are many sigma orbtials and they are interleved with the pi MOs.
- in your wiki present 5 pairs of comparative MOs from those you have visualised (benzene,borazine). Cover a range of bonding/antibonding characters and include both "sigma" and "pi" MOs.
- Note: "comparable" MOs can have different energies in each compound, so it is not sufficient to compare MOs simply based on energy ordering, you want to try and identify MOs by shape NOT energy or energy ordering.
- describe the MOs, compare and contrast differences and similarities and identify the overal MO character. See below for an example:
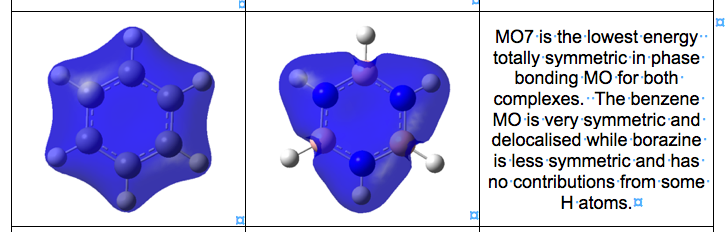
- discuss in your wiki (2-3 paragraphs) the concept of aromaticity, the simple ideas and also the more complex descriptions. You are required to address not just the simplistic models you have seen previously but to consider sigma and pi aromaticity.
- how do the real MOs relate to the common (very basic) conceptions of aromaticity?
- you are expected to explain why the concept of overlapping pz AOs is NOT a good description for aromaticity.
- you may find the introduction from this paper helpful.
- optional Comment on or analyse or describe some aspect of this system which you found interesting.
